Ann Arbor staging classification for Hodgkin and non-Hodgkin lymphomas
Stage I
Involvement of a single lymph node region (I) or of a single extralymphatic organ or site (IE)*
Stage II
Involvement of two or more lymph node regions or lymphatic structures on the same side of the diaphragm alone (II) or with involvement of limited, contiguous extralymphatic organ or tissue (IIE)
Stage III
Involvement of lymph node regions on both sides of the diaphragm (III) which may include the spleen (IIIS) or limited, contiguous extralymphatic organ or site (IIIE) or both (IIIES)
Stage IV
Diffuse or disseminated foci of involvement of one or more extralymphatic organs or tissues, with or without associated lymphatic involvement
Eastern cooperative oncology group (ECOG, Zubrod) performance scale
Performance status Definition

0 Fully active; no performance restrictions
1 Strenuous physical activity restricted; fully ambulatory and able to carry out light work
2 Capable of all selfcare but unable to carry out any work activities. Up about >50% of
waking hours
3 Capable of only limited selfcare; confined to bed or chair >50 percent of waking hours
4 Completely disabled; cannot carry out any selfcare; totally confined to bed or chair
Excerpted from Oken, MM, et al. Am J Clin Oncol 1982; 5:649.
International Workshop Criteria (IWC) for assessing response to treatment in non-Hodgkin's lymphoma (NHL)
Complete remission (CR)
Complete disappearance of all detectable clinical and radiographic evidence of disease
Disappearance of all disease-related symptoms if present before therapy
Normalization of biochemical abnormalities (eg, LDH) if definitely assignable to NHL
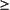
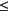
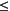
The spleen, if enlarged on CT scan prior to therapy, must have regressed in size and be non-palpable on physical examination
Complete remission unconfirmed (CRu)
Complete disappearance of all detectable clinical evidence of disease
Disappearance of all disease-related symptoms if present before therapy
Normalization of biochemical abnormalities
Residual lymph mass >1.5 cm in greatest transverse diameter that has regressed by more than 75 percent, or
Indeterminate bone marrow (increased number or size of aggregates without cytologic or architectural atypia)
Partial remission (PR)
At least 50 percent reduction in the sum of the product of the greatest diameters of the six largest dominant nodes or nodal masses (SPD)
No increase in the size of other nodes, liver, or spleen
No new sites of disease
Bone marrow biopsy results are not relevant for determination of PR in this system
Stable disease (SD)
Less than a partial remission but not progressive disease
Progressive disease (PD) [for patients with a prior PR or NR]
50% or more increase in the SPD from nadir of any previously identified abnormal node, or
Appearance of any new lesion during or at the end of therapy
Relapsed disease (RD) [for patients with a prior CR or CRu]
Appearance of any new lesion, or
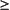
50% or more increase in the greatest diameter of any previously identified node >1 cm in short axis or in the SPD of more than one node
Excerpted from Cheson, BD, et al. J Clin Oncol 1999; 17:1244 and Juweid, ME, et al. J Clin Oncol 2005; 23:4652.
Risk factors for neuraxis involvement by non-Hodgkin's lymphoma




